Modeling & Simulation
Mathematical models can transform raw experimental data into actionable insight. I help clients apply binding, kinetic, and pharmacological modeling to understand mechanisms, design better experiments, and make informed decisions about therapeutic strategies. Whether the goal is exploring new modalities, de-risking dosing approaches, or simply interpreting complex datasets, I provide modeling support that complements experimental work.
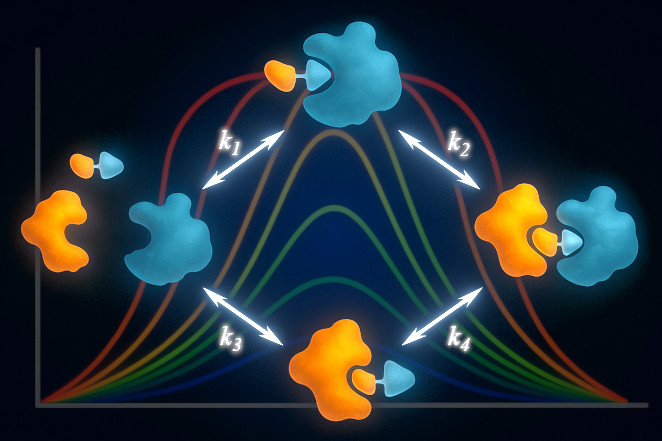
Binding & Kinetic Modeling
Quantitative models of protein-protein or protein-ligand interactions provide mechanistic clarity that goes beyond raw binding curves. I help clients build models that capture real biological complexity and guide decision-making.
Services include:
- Custom binding & kinetic models – develop equilibrium and kinetic models tailored to specific systems (e.g., receptors, enzymes, transporters, bivalent antibodies, bispecifics, ADCs, LYTACs, degraders, molecular glues, etc.)
- Mechanistic interpretation – fit models to experimental data to extract meaningful parameters such as affinities, rates, or effective concentrations
- Experimental design support – use modeling to refine assay conditions and predict outcomes before data are collected
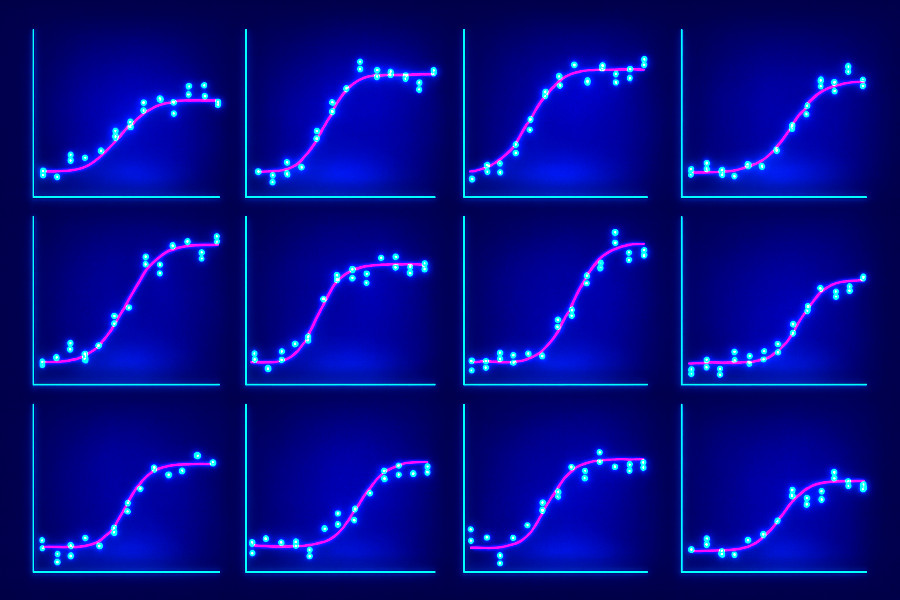
Data Analysis & Curve Fitting
Many projects require rigorous analysis of dose-response or kinetic data. I help clients fit data with the appropriate mathematical framework to extract reliable parameters.
Services include:
- Dose-response analysis – determine EC50/IC50 values, efficacy, cooperativity, and allosteric effects
- Kinetic curve fitting – apply appropriate models to time-course or binding kinetics data for parameter estimation
- Uncertainty and sensitivity analysis – identify which parameters are well constrained and which require further experimental data
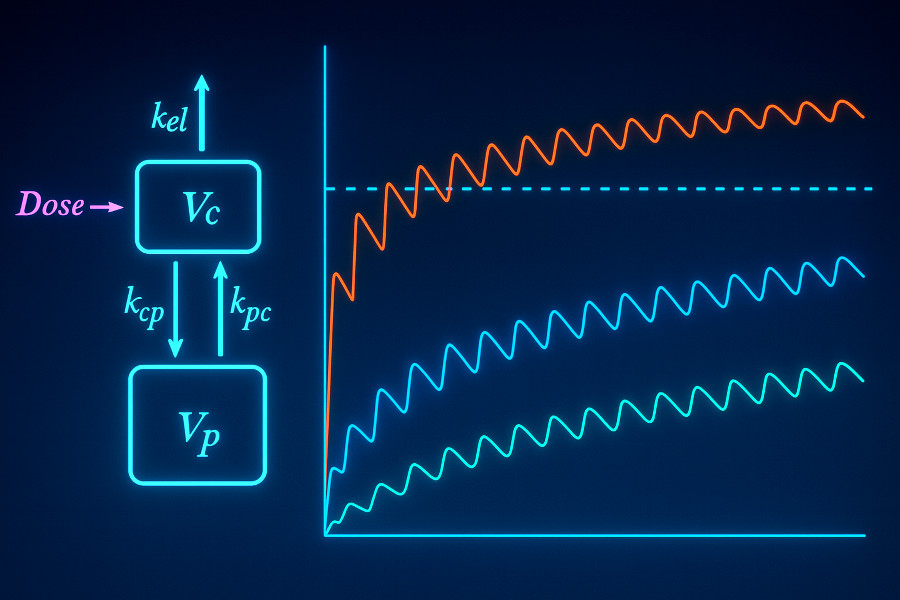
Pharmacokinetic / Pharmacodynamic (PK/PD) Modeling
Understanding the relationship between dose, exposure, and effect is critical for advancing therapeutic programs. I support clients in applying PK/PD models to inform development strategy.
Services include:
- PK/PD model development – apply standard or custom frameworks to small molecules or biologics
- Dose optimization – simulate dosing strategies to balance efficacy, safety, and therapeutic index
- Integration with mechanistic data – link PK/PD models to biochemical or binding data for a more complete picture of target engagement and response
- Scenario analysis for decision support – extend PK/PD models to explore “what if” cases (e.g., target engagement thresholds, modality comparisons, or dosing strategies) to provide strategic insight while highlighting key uncertainties
Why Partner with Me?
With experience bridging protein biochemistry, structural biology, and quantitative modeling, I provide clients with practical tools to connect data to mechanism. My support ranges from building models de novo to interpreting existing datasets, always with the goal of turning complexity into clear, actionable decisions.
Interested in working together? Contact me
